久々になりますが以下ご参考まで貼っておきます。
Excerpts are on our own.
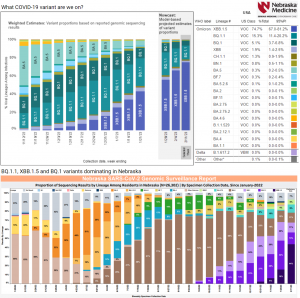
What COVID-19 variants are going around in February 2023? (02/15/2023) | @nebraskamed
Living Evidence – SARS-CoV-2 variants | @nswaci
SARS-CoV-2 variant rapid risk assessment report: XBB.1.5 (01/18/2023) | @NewsroomGC
excerpts – parts of a table

Expert insight: FAQ on COVID-19 subvariant XBB.1.5 (01/17/2023) | @SameerElsayedMD,@ConversationCA,@westernu
How likely is XBB.1.5 to cause serious illness?
…
Nonetheless, XBB.1.5 is perceived as being equally capable of causing serious illness in elderly and immunocompromised persons compared to previous Omicron subvariants of concern.
Are current mRNA vaccines effective against XBB.1.5?
XBB.1.5 and XBB.1 are the Omicron subvariants with the greatest immune-evasive properties. Therefore, one of the most contentious issues surrounding XBB.1.5 relates to the degree of protection afforded by currently available mRNA vaccines, including the latest bivalent booster formulations.
Researchers from the University of Texas determined that first-generation and bivalent mRNA booster vaccines containing BA.5 result in lacklustre neutralizing antibody responses against XBB.1.5. A report (yet to be peer reviewed) from investigators at the Cleveland Clinic found that bivalent vaccines demonstrate only modest (30 per cent) effectiveness in otherwise healthy non-elderly people when the variants in the vaccine match those circulating in the community.
Furthermore, some experts believe the administration of bivalent boosters for the prevention of COVID-19 illness in otherwise healthy young individuals is not medically justified nor cost-effective.
In contrast, public health experts from Atlanta, Ga. and Stanford, Calif. reported that although the neutralizing antibody activity of bivalent booster vaccines against XBB.1.5 is 12 to 26 times less than antibody activity against the wild-type (original) SARS-CoV-2 virus, bivalent vaccines still perform better than monovalent vaccines against XBB.1.5.
However, investigators from Columbia University in New York found that neutralizing antibody levels following bivalent boosting were up to 155–fold lower against XBB.1.5 compared to levels against the wild-type virus following monovalent boosting.
This suggests that neither monovalent nor bivalent booster vaccines can be relied upon to provide adequate protection against XBB.1.5.
How can you protect yourself against XBB.1.5?
The rapid evolution of SARS-CoV-2 continues to pose a challenge for the management of COVID-19 illness using available preventive and therapeutic agents. Of note, all currently available monoclonal antibodies targeting the spike protein of SARS-CoV-2 are deemed to be ineffective against XBB.1.5.
Antiviral medicines such as remdesivir and Paxlovid may be considered for the treatment of eligible infected patients at high risk of progressing to severe disease.
Standard infection control precautions including indoor masking, social distancing and frequent handwashing are effective measures that can be employed for personal and population protection against XBB.1.5 and other subvariants of concern.
Although bivalent boosters may be considered for elderly, immunocompromised and other risk-averse individuals, their effectiveness in preventing COVID-19 illness due to XBB.1.5 remains uncertain.
Why is XBB.1.5 nicknamed ‘Kraken’?
…
COVID-19 Variants XBB and XBB.1.5: What You Should Know (02/07/2023) | @ReeveFoundation
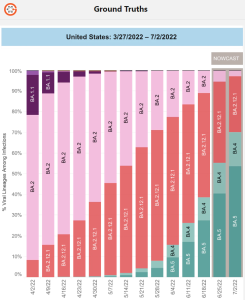
What is BA.2?
… BA.2 is a sub-lineage of Omicron’s BA.1 (BA.1.1.529, BA.1.1, and BA.3). …(CDC) COVID-19 Data Tracker reported that approximately 74% of new cases were linked to the BA.2 subvariant. During the height of BA.2 new cases, WHO stated that the BA.2 subvariant was easier to transmit from one person to another than its “sister” subvariant BA.1. …
The BA.2 subvariant is known as the stealth Omicron variant because its genetic mutations make distinguishing it from the Delta variant difficult. Doctors, public health, and infectious disease officials noted that while BA.2 is more transmissible than BA.1, its symptoms are not more severe.
XBB
While new cases of BA.5 – and its subvariants BQ.1, and BQ.1.1, an Omicron subvariant, were reported in Europe and America, cases of XBB were increasing in other parts of the world. … There is, however, early evidence pointing at a higher reinfection risk than other circulating Omicron sublineages.”
Infection Risk of XBB
… early findings show XBB may have a higher infection rate than previous Omicron subvariants. Experts continue to study XBB’s reinfection rate.
Transmission Risk of XBB.1.5
The risk of being infected by XBB.1.5 is approximately 40% higher than other Omicron subvariants. People infected have a higher chance of passing on the virus to others. Further affecting the risk of becoming infected with XBB.1.5 is its ability to slip past previous defense measures. … XBB.1 was 63 times less likely to be neutralized by antibodies found in vaccinated and infected people than the BA.2 subvariant and 49 times less likely to be neutralized when compared with BA.4 or BA.5. Meaning these subvariants can evade vaccines and antibodies built up in your body from a previous COVID-19 infection. …
Prevention
Despite the increased risk of infection from XBB.1.5, you can still take steps to reduce your risk of infection or the effects of XBB.1.5. The CDC recommends people wear masks when indoors, especially in poorly ventilated or crowded places. The agency also has vaccines and booster shots guidelines on its COVID-19 page.
COVID Behaviors Dashboard | @JohnsHopkinsCCP
XBB.1.5: What you need to know about COVID-19’s ‘Kraken’ variant (01/17/2023) | @UCDavisHealth
Omicron BQ and XBB subvariants most resistant viruses to monoclonal antibodies (02/13/2023) | @NewsMedical
XBB.1.5 Omicron subvariant: Questions patients may have (02/02/2023) | @SaraTheIceBerg,@AmerMedicalAssn
Covid-19: What do we know about XBB.1.5 and should we be worried? | @emahase_,@BMJ_latest
Early Estimates of Bivalent mRNA Booster Dose Vaccine Effectiveness in Preventing Symptomatic SARS-CoV-2 Infection Attributable to Omicron BA.5– and XBB/XBB.1.5–Related Sublineages Among Immunocompetent Adults (02/03/2023) | @CDCgov
Bivalent Covid-19 Vaccines — A Cautionary Tale | @DrPaulOffit,@NEJM